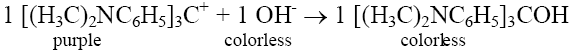
Crystal violet – hydroxide ion reaction rate law determination
Chemical kinetics is the study of the rates of chemical reactions. The rate of a chemical reaction describes how fast a reaction takes place, or in other words, how fast the reactant and product concentrations change. Factors such as reactant concentration, temperature, and presence of a catalyst effect reaction rate.
In this experiment, we examine the effects of changes in initial reactant concentration on reaction rate. Usually, an increase in reactant concentration causes an increase in reaction rate. This is because the more reactant molecules that are in a given volume of reaction mixture, the greater the likelihood that the molecules will collide and react. The purpose of the experiment is to determine the complete (including k with units) rate law for the reaction.
The
specific reaction rate we study is this experiments that of crystal
violet dye,
[(H3C)2NC6H5]3C+
and hydroxide ion, 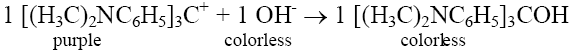
Crystal violet is purple, whilst both
We use a rate law to describe the dependence of a reaction rate on the corresponding reactant concentration. For the general reaction of two reactants, R and Q to form product P (R + Q --> P) we define its rate law as
rate
= k [R]x[Q]y
The proportionality constant, k, is called the rate constant, [R], and [Q] are the molar concentrations of R and Q, x and y are the orders in respect to the two reactants.
The order with respect to a reactant quantifies the effect of charges in the concentration of just that reactant on the reaction rate. They are not necessarily the coefficients in the balanced chemical equation.
To use the graphic method for determining reaction order with respect to a specific reactant, we must plot changes in the concentration of that reactant against time. We begin by measuring the concentration of that reactant, [R], during the course of a controlled reaction in which we keep all factors as constant as possible. One technique for achieving this consistency is to use a large excess of the other reactant, Q. During the course of the reaction, only a relatively tiny portion of the excess reactant Q will be consumed, so its concentration will remain relatively constant. Thus, any changes in the reaction rate will be due only to the decreasing concentration of R.
Once we have obtained the concentration data for reactant R under such controlled conditions, we plot the data, using the following graphics treatments, no more than one of which can yield a straight line graph: [R] versus time (true if the reactions is zeroth order in R), ln[R] vs time (straight of 1st order in R), or 1/[R] vs. time(straight of 2nd order in R). Remember, Beer's law coorelates Abs and concentration (A =ebc) Knowing e = 5.0 x 104 L/(cm mol) and b = 1 cm for this experiment, you can convert (or have Excel do it) each Abs measurement into concentration. We can then simply plot [] vs. time, ln [] vs. time, and 1/[] vs. time. Hopefully one of them will be linear and we will have the order of reaction for crystal violet determined.
So this graphical
method procedure works swimmingly for determining the order of reaction
for the
colored reactant, crystal violet. But what about the order of reaction
for the
We will need to know the rate of reaction of the 0.100 M run and
the rate of reaction of the 0.05 M reaction. The slope of the [] vs.
time line is in fact the reaction rate. So we just conmpare those
slopes, and holefully they will be something convient in comparison to
one another (the same, double, 4x, whatever)
In this
experiment, we investigate the rate of reaction between crystal violet
and
In the
first trial, we will mix a crystal violet solution with an
We will then
do a second trial, changing only the initial
1. Pick out your spectrometer and get it set up. Calibrate, set for 1 reading/second and collect data for 240 seconds.
2. Using a volumetric pipet, put 5.00 mL of 0.050 M NaOH solution in a clean dry test tibe. Obtain 5.00 mL of 1.5 x 10-5 M crystal violet into a second dry test tube.
4. To begin the reaction, pour the samples of crystal violet and NaOH together and pour back and forth 3 times (to mix). Start you timer immediately upon first mixing.
5. After the solution is mixed, rinse a cuvet twice with a small amount of the reaction mixture. Then fill the cuvet and dry the outside.
6. Place the cuvet in the spectrometer and start the Abs-time readings at exactly 90 seconds past the time the solutions were mixed.
7. After the run, process the saved data as explained on the data sheet.
8. Repeat steps 2 through 7 with 5.0 mL of 0.100 M NaOH and 5.0 mL of 1.5 x 10-5 M crystal violet.
10. Open a data file in MS Excel, create 1 graph containing three sets of data, A vs. time, Ln A vs. time, 1/A vs. time for the lower concentration run (0.050 M NaOH) . Make sure you add a trend line and R2 value to each line. They should all be well labeled. A second graph of just A vs. time is needed for the second (0.100M NaOH) run.
1. Write a very brief summary of the
entire procedure. It should be less than 5 sentences.
2. The following data were obtained for the reaction: 2 NO2(g)-->2 NO(g)
+ 1 O2(g)
| time (s) | [NO2] M | ln [NO2] | 1/[NO2] |
| 0 | 0.8333 |
|
|
| 20 | 0.4167 |
|
|
| 40 | 0.2778 |
|
|
| 60 | 0.2083 |
|
|
a. Determine the ln [NO2]
and 1/[NO2] for each data point.
b. Make a computer generated graph of [NO2] vs
time, ln [NO2] vs time, and 1/[NO2]
vs time. (one graph, 3 lines) Determine the slope and R2 value of
each line. Here is a screencast
showing you how to do the graphs.
c. Which graph yields the best straight line (R2
value closest to 1) ?
d. What is the reaction order with respect to NO2?
e. Write the rate law for this reaction including the numerical value
for k.
3. Come up with a data sheet for the appropriate needed data. Show your
instructor on the way into lab.
4. Bring your portable memory drive (flash drive thingie) to extract data from lab.